[ad_1]
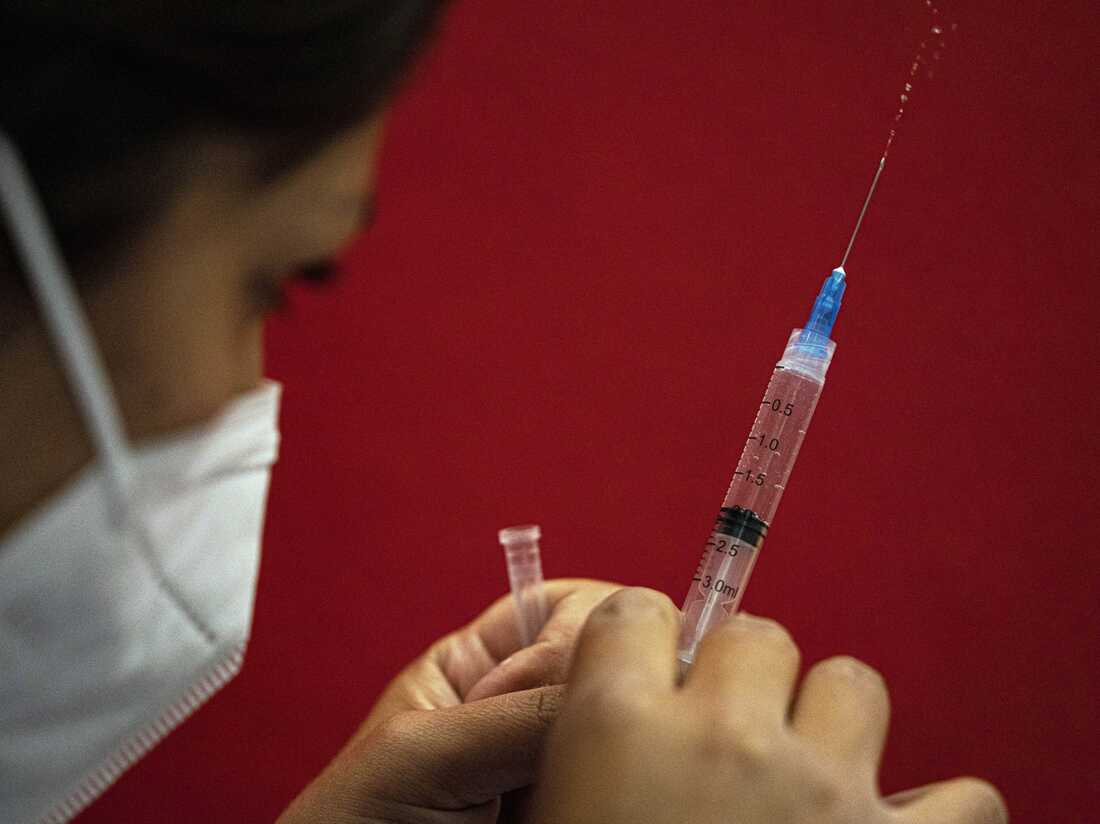
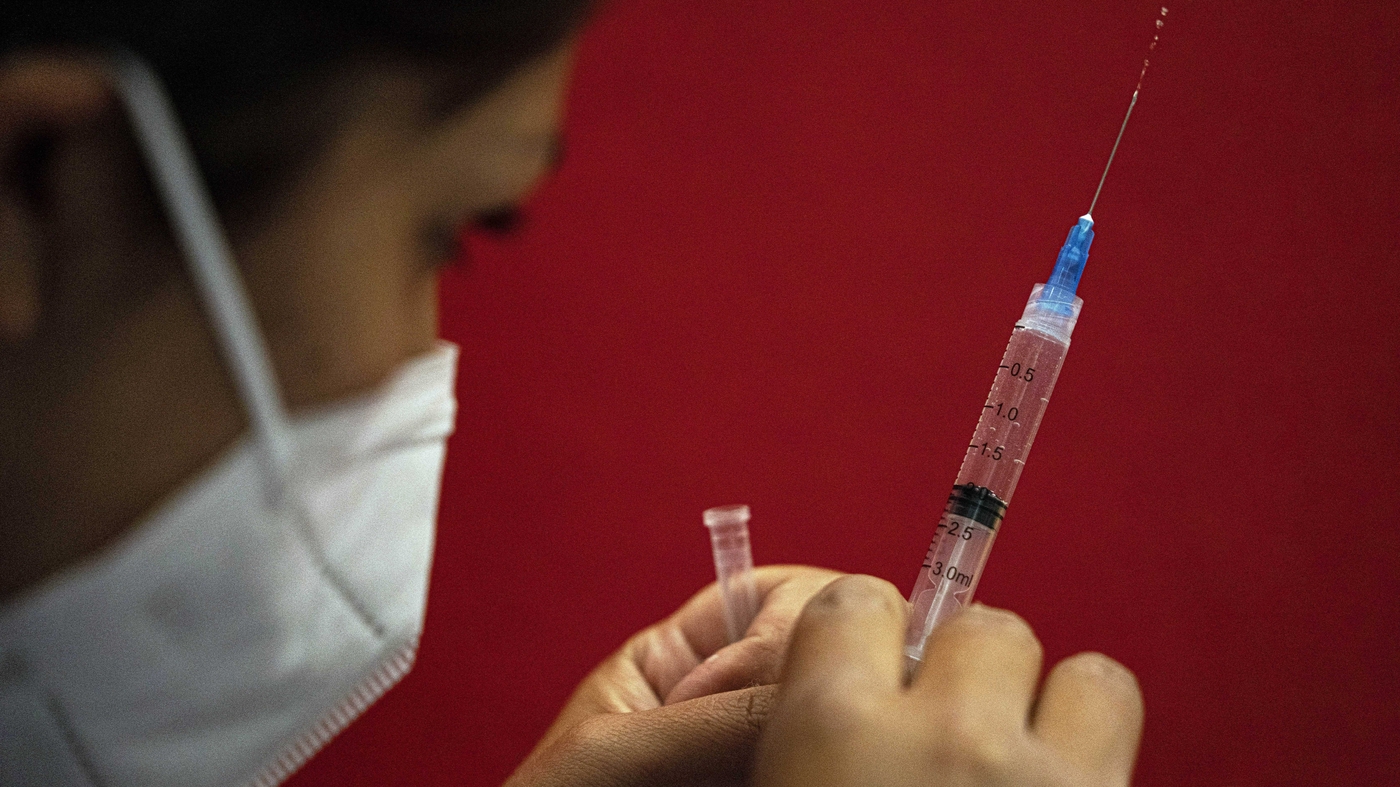
The COVID-19 vaccine will be up to date in hopes of concentrating on the strains of omicron that will be circulating later this year.
Esteban Felix/AP
cover caption
toggle caption
Esteban Felix/AP

The COVID-19 vaccine will be current in hopes of targeting the strains of omicron that will be circulating later this calendar year.
Esteban Felix/AP
A panel of expert advisers to the Food and Drug Administration voted unanimously Thursday to propose that the COVID-19 vaccine be current to focus on rising subvariants of omicron.
The COVID shot which is presently offered is acknowledged as a “bivalent” vaccine mainly because it was personalized to focus on both equally the original strain of the coronavirus and the omicron subvariants that dominated last winter.
But the Fda panel advisable that drugmakers abandon the bivalent style and rather move to a “monovalent” vaccine that only targets omicron subvariants. The notion is to roll out the freshly formulated photographs in anticipation of a doable uptick in conditions this tumble.
The committee especially supported concentrating on the subvariant XBB.1.5, which accounts for about 40% of new infections in the U.S.
In an examination, Food and drug administration scientists explained details from vaccine manufacturers show that an current monovalent formulation that targets XBB subvariants “elicits more powerful neutralizing antibody responses” versus XBB strains than recent bivalent vaccines.
“There won’t look to be any certain advantage to a bivalent vaccine,” said Dr. Eric Rubin, a professor of immunology and infectious ailments at Harvard who is also a member of the advisory committee.
While there was wide agreement about shifting to a monovalent vaccine, there was substantial discussion between committee users about no matter if the COVID-19 vaccine must be dealt with like the influenza vaccine, which is revamped every single year in anticipation of flu period.
“People recognize a annually influenza vaccine,” mentioned Dr. Peter Marks, director of the FDA’s Centre for Biologics Evaluation and Study. “At this stage it may not be annually, but, for all intents and purposes, it looks like by subsequent fall there will be further drift from this [strain] and we might have to arrive back right here.”
But some anxious that drawing much too shut a parallel to influezna could truly direct to confusion amid People in america.
“This is not the flu,” mentioned committee member Dr. Paul Offit, a professor of pediatrics at the Children’s Healthcare facility of Philadelphia. He pointed out that several People by now have some protection from extreme disease from COVID-19 because of prior an infection, vaccination, or both.
“I’m all for updating this vaccine, but I think we want to outline… who seriously rewards from booster dosing? Simply because it can be not all people,” he stated.
It’s not nevertheless apparent who federal officers will advocate need to get the up-to-date omicron vaccine.
Throughout the assembly, the CDC shared info that exhibits that, since final April, COVID hospitalizations and fatalities have been lower in most teams. But they have been disproportionately higher in folks who are 75 or older, suggesting this group may possibly need additional safety. Those people with wellbeing concerns like continual lung condition or diabetes might also have larger pitfalls.
The Fda is now likely to take into consideration the committee’s dialogue and will possible situation an formal advice about the vaccine formulation in a couple of times, which will give vaccine makers a route to stick to.
If all goes in accordance to program, it’s expected the new vaccines must be out in the tumble – by around late September or early October.
[ad_2]
Resource website link