[ad_1]
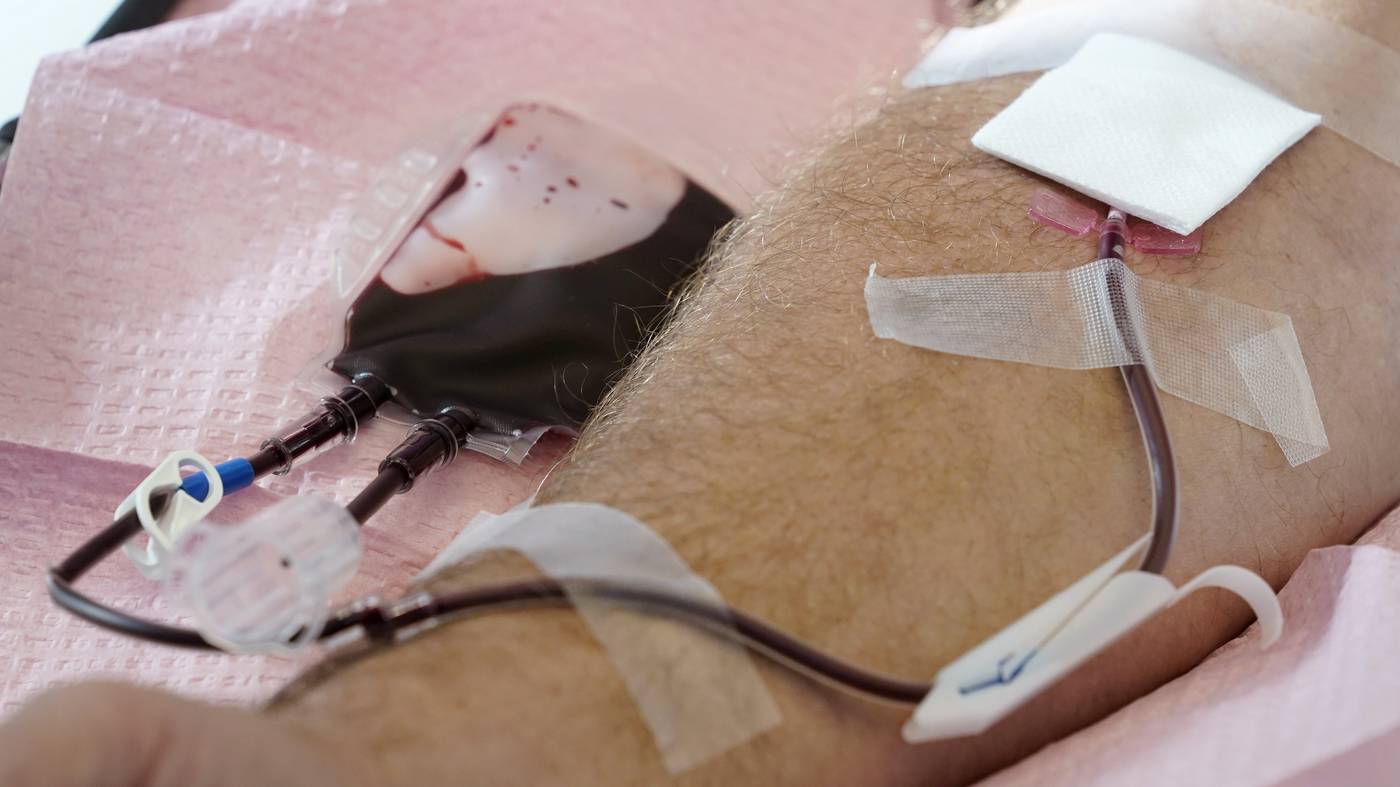
The U.S. is going to relieve restrictions on blood donations from homosexual and bisexual men and other groups that customarily experience greater risks of HIV. Below, tubes immediate blood from a donor into a bag in Davenport, Iowa, on Friday, Nov. 11, 2022.
Charlie Neibergall/AP
hide caption
toggle caption
Charlie Neibergall/AP
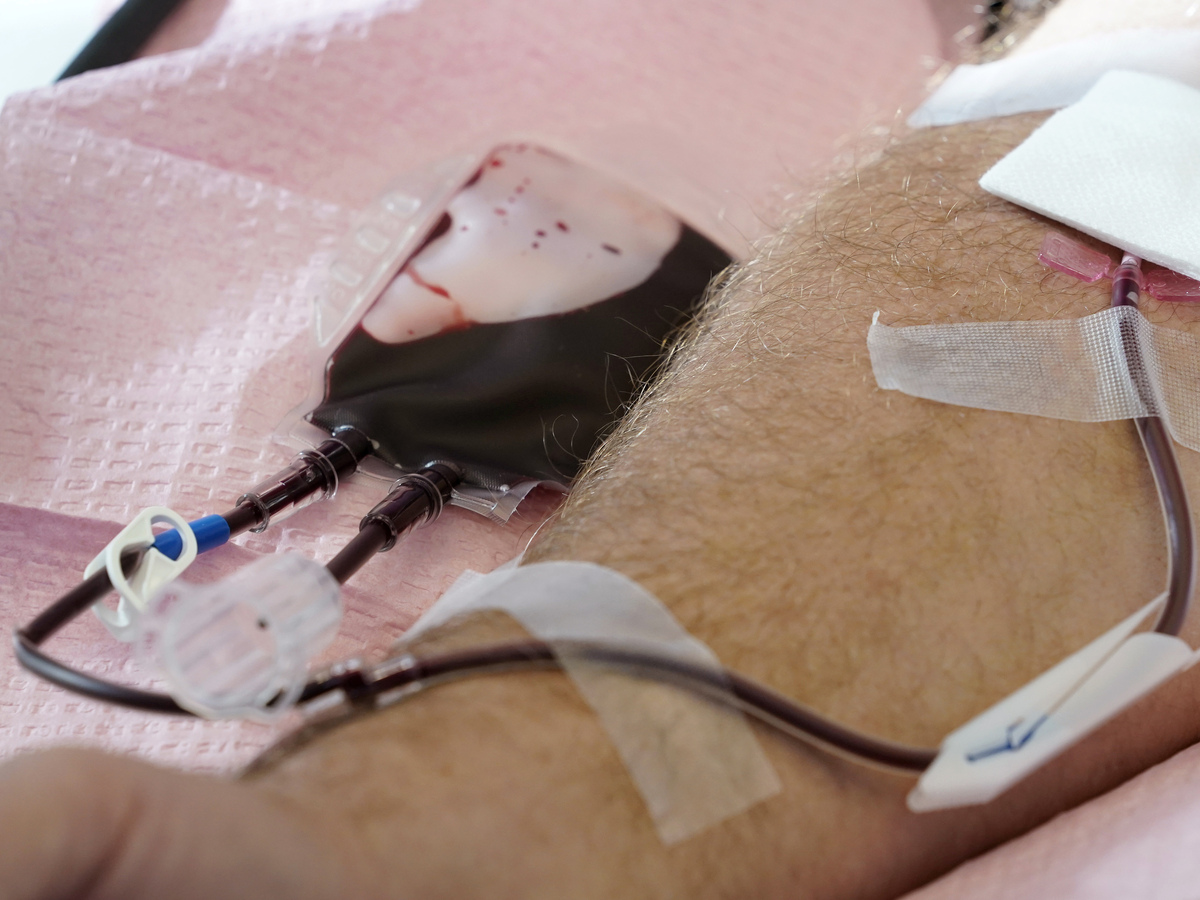
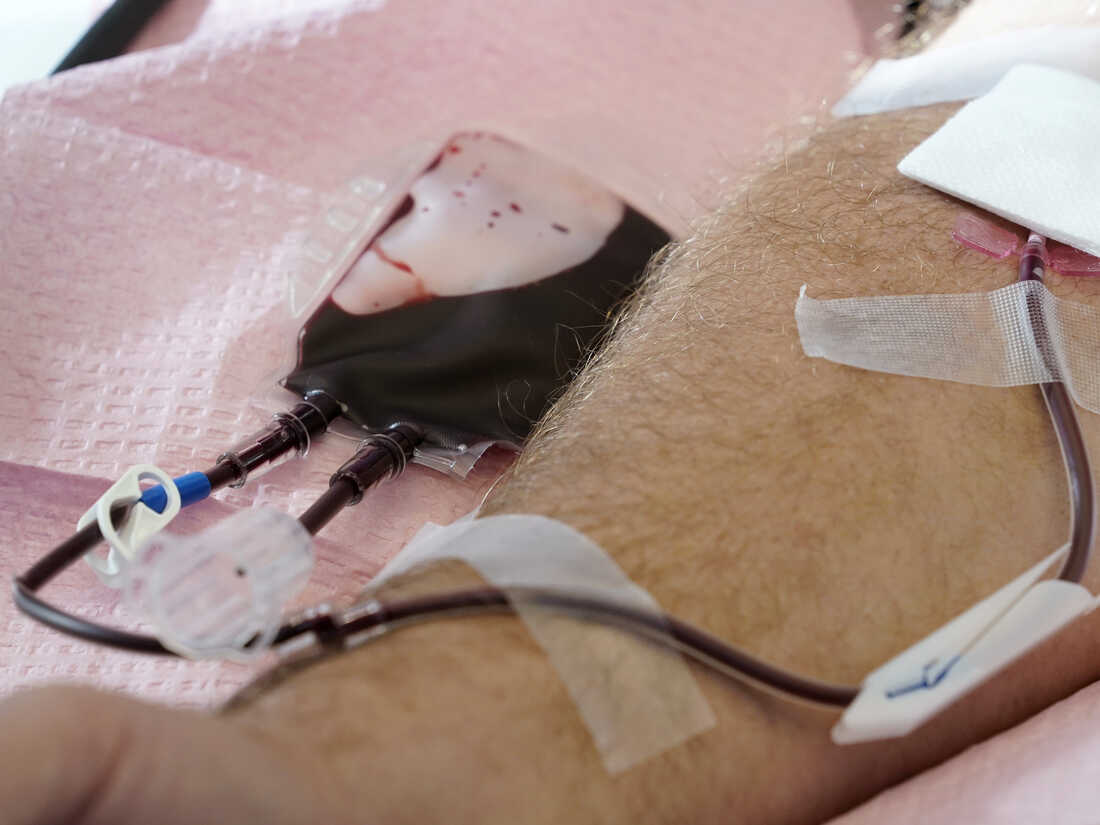
The U.S. is shifting to ease restrictions on blood donations from homosexual and bisexual adult males and other teams that ordinarily facial area higher hazards of HIV. Here, tubes immediate blood from a donor into a bag in Davenport, Iowa, on Friday, Nov. 11, 2022.
Charlie Neibergall/AP
The Foods and Drug Administration (Fda) reported Thursday it had officially eradicated constraints that experienced formerly prohibited many blood donations by gay and bisexual guys — a longstanding coverage that critics say is discriminatory.
In a information release, the federal company reported it will advise a collection of “particular person threat-primarily based queries” that will be the very same for just about every blood donor, irrespective of their sexual orientation, gender or intercourse. Those who have had anal sex with a new sexual companion, or much more than 1 sexual husband or wife, inside of the previous three months would be questioned to hold out to donate blood.
“The implementation of these suggestions will represent a important milestone for the company and the LGBTQI+ group,” Dr. Peter Marks, director of the FDA’s Centre for Biologics Evaluation and Investigate, claimed in the launch.
With the up to date guidelines, most homosexual and bisexual males who are in a monogamous relationship with a gentleman will no for a longer period have to chorus from sex in order to donate blood.
Beforehand, Food and drug administration guidelines for donating blood — which have been past up to date in 2020 — said that guys who have sexual intercourse with men are allowed to donate blood soon after a a few-month deferral time period in which they chorus from having intercourse with yet another male.
Though the selection of persons eligible to donate blood has expanded, the agency claimed it will proceed to keep an eye on the basic safety of the blood offer.
The 40-year-old constraints have been to secure the blood source from HIV
Limits on donating blood date back to the early days of the AIDS epidemic and were designed to defend the blood provide from HIV.
At very first, gay and bisexual men were entirely prohibited from donating blood. But more than time, the Fda in the end calm the lifetime ban. Having said that, the company continue to retained some restrictions in location.
The newly up-to-date tips are aimed at addressing a long time-extended criticisms that the prior policy was discriminatory and out-of-date, and posed nevertheless another barrier to bolstering the nation’s blood supply.
Blood banking institutions by now routinely screen donated blood for HIV.
And for a long time, organizations which includes the American Health care Association, the American Red Cross and various LGBTQ+ advocacy groups have identified as for a rule transform.
The variations are a largely welcome shift in a new way, advocates say
Response to the news has been mostly good from advocates, clinical teams and blood banking institutions.
“This shift towards particular person donor assessments prioritizes the protection of America’s blood provide when treating all donors with the fairness and respect they are worthy of,” mentioned Kate Fry, CEO of America’s Blood Centers, a non-revenue firm that delivers collectively group-based mostly and independent blood facilities, in a assertion.
Fry reported that the FDA’s last direction is based on information that shows the ideal security from illnesses, like HIV, is by way of potent testing of all blood donation — and a uniform screening method for each individual donor.
President and CEO of GLAAD Sarah Kate Ellis echoed that approval in a assertion, declaring “The FDA’s selection to stick to science and issue new recommendations for all People in america, irrespective of sexual orientation, who selflessly donate blood to assist help you save lives, indicators the beginning of the close of a darkish and discriminatory previous rooted in concern and homophobia,”
On the other hand, Ellis stated although the new steerage is a step in the right route, there is nonetheless a barrier for LGBTQ+ men and women who are on PrEP, an Fda-accepted drug confirmed to protect against the transmission of HIV, who could want to donate blood.
“GLAAD urges the Food and drug administration to continue on to prioritize science about stigma and deal with all donors and all blood equally,” she added.
NPR’s Rob Stein and Will Stone contributed to this report.
[ad_2]
Supply connection